Research Application Technology Center
Abstract
This study investigates the structural dimensions of Bacteriophage Lambda using the non-contact mode of Atomic Force Microscopy (AFM). Bacteriophage Lambda, a virus that infects Escherichia coli, is widely used in molecular biology and genetics. The non-contact mode of AFM is particularly suitable for measuring delicate biological samples without damaging the surface. Our results demonstrate the effectiveness of this method in obtaining accurate topographical information of Bacteriophage Lambda, highlighting its potential applications in nanotechnology and biological research.
Introduction
Bacteriophage lambda (λ phage) is a double-stranded DNA virus that specifically infects Escherichia coli (E. coli) and has played a central role in the development of molecular biology and genetics.1 Discovered by Esther Lederberg in the early 1950s2, λ phage is particularly renowned for its ability to toggle between two distinct life cycles: the lytic and lysogenic pathways. In the lytic cycle, the phage commandeers the bacterial host’s machinery to replicate its genome and assemble new virions, eventually leading to cell lysis. Alternatively, during the lysogenic cycle, the phage genome integrates into the bacterial chromosome and remains dormant as a prophage until environmental stress triggers reactivation of the lytic program.3
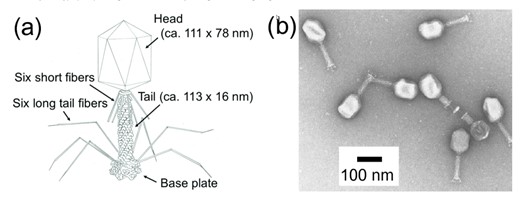
Figure 16. (a) Schematic image of Bacteriophage Lambda. (b) TEM image of bacteriophage Lambda using negative staining reagents.
Structurally, as figure 1 above, λ phage consists of an icosahedral capsid enclosing its DNA genome, a contractile tail, and tail fibers that mediate host recognition and DNA injection into the bacterial cytoplasm.4 Owing to its capacity to carry foreign DNA—up to approximately 20 kilobases—λ phage has been extensively used as a cloning vector in genetic engineering. This has enabled researchers to study gene function and regulation, construct genomic libraries, and express recombinant proteins efficiently.5
Bacteriophage lambda has a variety of applications in research beyond its use as a cloning vector. Here are some notable examples:
Phage Display
Lambda phage can be used to display foreign peptides or proteins on its coat proteins, enabling the study of protein–protein interactions and facilitating the discovery of drug and vaccine candidates.
Gene Transfer
Engineered lambda phage can serve as a gene delivery vehicle to bacterial cells, allowing for controlled studies of gene function and regulatory mechanisms.
Vaccine Delivery
By displaying antigens on the phage surface, lambda phage can be used as a platform to enhance immune responses, offering potential for vaccine development.
Bio-detection
Lambda phage expressing reporter genes can be used in biosensors to detect specific bacterial targets with high sensitivity and specificity.
Bio-control
Lambda phage may be employed as a biological control agent to selectively eliminate bacterial pathogens, particularly in agricultural and food safety applications.
Nanomedicine
As a potential nanocarrier, lambda phage is being explored for targeted delivery of therapeutic agents, including drugs and genetic material, in precision medicine.
In this study, the structure of Bacteriophage Lambda is measured using the non-contact mode of the AFM system. The non-contact mode is particularly effective for measuring very soft and delicate samples such as polymers, cells, and viruses. This technique is widely used in material science, nanotechnology, and biology to study the topographical information and properties of surfaces at the nanoscale.
Preparation and method
To prepare a sample for non-contact mode AFM imaging of Bacteriophage lambda, start by freshly cleaving a mica surface to make it very smooth and clean. Then, apply a magnesium ion (Mg²⁺) solution to the surface and let it sit for a short time. This helps the negatively charged phage particles stick to the mica. After removing the extra solution, place a drop of the purified lambda phage solution onto the treated mica and allow it to dry completely in air. Because non-contact mode detects the surface without physical contact, The sample should be firmly secured and positioned flat to prevent movement or distortion during scanning. The flatness and cleanliness of mica, combined with proper ion treatment, are essential for obtaining stable, high-resolution images of the phage structure in non-contact mode. Imaging was performed using the NX10 system in non-contact mode, equipped with a PPP-FMR cantilever, which provides high sensitivity and is well-suited for capturing the fine surface features of phage particles without damaging them.
Results
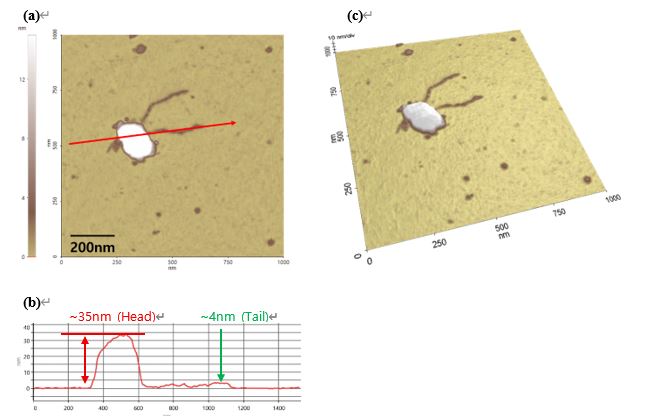
In this article, the structure of Bacteriophage lambda is well measured with AFM system (figure 2). Since the Bacteriophage has very delicate surface itself, to get real topographical information without any damage, non-contact mode is one of the most effective ways to measure. Furthermore, it is already complicated to prepare the Bacteriophage on MICA. So, it is recommended to use AFM since it has less preparation before loading the sample on the tool compared to another tools such as SEM or TEM which need additional environmental conditions like vacuum system. And it does not need any treatment on sample like conductive coating which means researchers can observe the sample with very native state. Furthermore, users can see the specific 3D structure of Bacteriophage lambda.
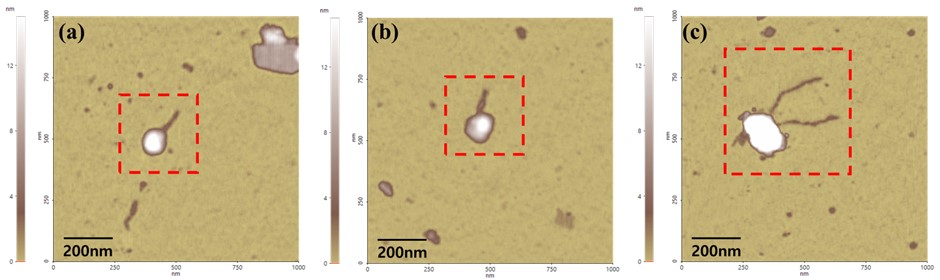
Figure. 2 The Bacteriophage Lambda images measured by AFM (Non-contact mode)
Using non-contact mode, we observed the typical structure of Lambda Phage. The head was measured to be approximately 100 nm in width and 20 nm in height. Figure 3(a) and Figure 3(b) exhibit the dimensions of bacteriophage lambda. The tail was observed to be about 4 nm in height and between 200 nm and 500 nm in length. However, the tail fibers were not detected during the measurements. Several factors could contribute to the common purification procedures used for structural protein analysis such as PEG precipitation, gradient centrifugation, and ghost particle preparation. Those can impose mechanical and chemical stress on bacteriophages, potentially leading to structural damage. Notably, delicate external features like tail fibers may be particularly vulnerable during these steps. Therefore, the observed loss of tail fibers in AFM imaging may not solely result from weak substrate adhesion but could also stem from damage introduced during the purification or handling process. These findings support the importance of optimizing purification protocols when preserving morphological integrity is critical for downstream structural analysis.7
The AFM system provides not only 2D images but also line profiles and 3D rendering images. They are also shown in Figure 3(c).
Figure. 3 (a), (b) Height image and its line profile and (c) 3D image of Bacteriophage Lambda
Conclusion
In this study, the dimensions of Bacteriophage Lambda particles were successfully measured. Using the non-contact mode of Atomic Force Microscopy (AFM). The delicate and fragile surface of Bacteriophage Lambda necessitates a non-destructive method for observation, making AFM an ideal tool. The non-contact mode of AFM allows for the measurement of the sample surface without causing any damage, which is particularly advantageous for biological samples. AFM preparation does not require vacuuming or coating, enabling the examination of biological samples in their natural state.
References
1. Ptashne, M. (2004). A Genetic Switch: Phage Lambda Revisited. 3rd ed. Cold Spring Harbor Laboratory Press.
2. Lederberg, E. M. (1951). Lysogenicity in E. coli K-12. Genetics, 36(6), 560–561.
3. Oppenheim, A. B., Kobiler, O., Stavans, J., Court, D. L., & Adhya, S. (2005). Switches in bacteriophage λ development. Annual Review of Genetics, 39, 409–429.
4. Casjens, S. R., & Hendrix, R. W. (2015). Bacteriophage lambda: Early pioneer and still relevant. Virology, 479–480, 310–330.
5. Sambrook, J., & Russell, D. W. (2001). Molecular Cloning: A Laboratory Manual. 3rd ed. Cold Spring Harbor Laboratory Press.
6. Sahiro. K., Kawato. Y., Koike. K. & Sano. T. (2022) Preyssler-type phosphotungstate is a new family of negative-staining reagents for the TEM observation of viruses
7. Boulanger, P. (2009). Purification of Bacteriophages and SDS-PAGE Analysis of Phage Structural Proteins from Ghost Particles.
Application
Related Contents

Solving Thin-Film Uniformity Challenges on Curved Surfaces with Imaging Spectroscopic Ellipsometry

AFM Augmented Sample Fabrication for Next-Generation Layered Materials Devices